Live-Saving Technology
Patented innovations at the intersection of mineral science and medicine, designed to save lives in critical situations.
Is this Rocket Sience? It's definitely Rock Science!
Clay minerals have been known for decades for their hemostatic effect. In the early 2000s, scientists have discovered Halloysite Nano Tubes (HNT) – a powerful clay mineral for use in a variety of purposes.
Halloysite is a naturally occurring mineral, formed into a tubular shape by geothermal effects and achieves an enormously large morphological surface. This structure in combination with a favorable charge ratio for blood clotting makes Halloysite the rock star among clay minerals.




High-Performance Carrier Material and precise processes for high-performance hemostasis
The use of a high-pressure hydroentaglement process creates a homogeneous, interwoven textile surface structure with high tear resistance in the longitudinal direction. The hydrophilic perforated matrix structure of the gauze has interconnected pores and channels. This allows a large amount of water to be absorbed from the blood, leaving concentrated blood components at the bleeding site to promote hemostasis. In addition, the gauze optimizes pressure transfer to the wound bed, thereby supporting mechanical hemostasis.
For medical purposes a high purity material is essential. Transforming Halloysite into a medically usable material requires a complex and highly controlled process to ensure the safety and effectiveness of our solutions for life science applications. Speed Care Mineral specializes in mineral processing and refinement.
A precision printing process was developed to apply the halloysite mineral to the carrier material. Each production batch is tested to ensure rapid hemostasis, and the detachment behavior is examined to confirm that no particles can enter the bloodstream.


Physical mode of action
The adhesion and local concentration of platelets in the event of bleeding is accelerated due to the electrostatic attraction between the negative outer shell of the Halloysite Nanotubes (HNT) and the positive surface of the platelets. It is therefore a physical effect.
The tubular structure of Halloysite allows water to be stored inside. In contact with blood, the water is removed and platelets are concentrated outside the tubes. Platelets are responsible for the primary natural hemostasis in case of an injury. The local concentration of platelets simplifies and accelerates their adhesion and aggregation. In the final stage, a (white) thrombus develops, which ideally seals the vascular defect. 1
Surface Interaction: Negatively charged nanotube surfaces modulate local platelet accumulation through electrostatic interactions and protein-mediated binding.
Mechanical Support: Additionally, the gauze structure transmits pressure, stabilizing the wound site.
Fluid absorption: Capillary-driven fluid uptake into halloysite nanotubes and parallel absorption by porous, hydrophilic gauze concentrate blood components locally.
Clot Formation: Localized clot formation supports sealing of the vascular defect.
This life-saving technology enables highly effective and safe hemostasis, allowing even severe bleeding to be controlled quickly and reliably.
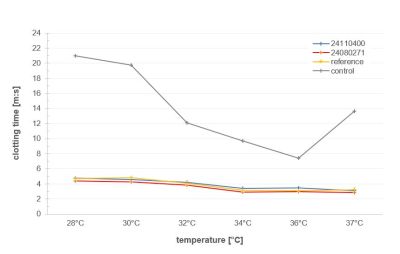
Evaluation of hemostatic performance of SpeedM® under hypothermic conditions.
The trauma triad of death is a medical term describing the combination of hypothermia, acidosis, and coagulopathy. This combination is commonly seen in patients who have sustained severe traumatic injuries and results in a significant rise in the mortality rate. Hypothermia impairs coagulation: the blood clots more slowly, stable clots form later, and bleeding lasts longer. In an intensive care study, the risk of death approximately doubled when patients arrived at the intensive care unit hypothermic.
A study investigated the thermal stability of the hemostatic performance of SpeedM® to determine the clotting time in human citrated plasma under hypothermic conditions.
All test samples and the reference demonstrated substantially shorter clotting times versus the control group at all temperatures.
In vitro studies with recalcified citrate plasma show that SpeedM® significantly shortens clotting times compared to the control over a hypothermia-simulating temperature range of 28–37 °C.
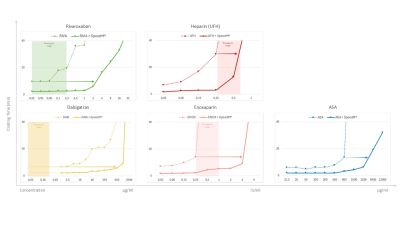
Hemostatic performance of SpeedM® under anticoagulated conditions: an in vitro study.
Uncontrolled bleeding remains one of the most common preventable causes of death following trauma. Rapid and effective hemostasis is therefore essential to sustainably reduce morbidity and mortality. This challenge is further amplified by the increasing number of patients receiving long-term anticoagulant therapy. As a result, an increasing proportion of trauma patients may present while taking such medications, making bleeding control more difficult and time-critical.
This study evaluated the hemostatic performance of SpeedM® in human citrated plasma under defined anticoagulant exposure by comparing clotting time with and without the dressing and estimated therapeutic reserve from the isoeffective rightward shift of anticoagulant concentration–response curves.
In the presence of the SpeedM® hemostatic dressing, the clotting time was consistently shorter for all anticoagulants tested than in anticoagulated plasma without dressing, indicating preserved procoagulant activity in this plasma-based model.
SpeedM® shows effective hemostatic performance under anticoagulated conditions: in a standardized in vitro plasma model, it consistently shortened clotting time across multiple anticoagulant classes and maintained this effect even at higher anticoagulant doses, indicating a functional safety margin.
High Tech Development – Easy to use.
Tactility: The SpeedM® carrier matrix is neither too soft nor too stiff. On the one hand, it must be possible to palpate injured blood vessels in wounds - on the other hand, the material must be firm enough to allow for precise placement in deep wound areas. Numerous user surveys and 29 development steps were necessary to achieve the current result.
Drainage: The carrier matrix features targeted perforations created by a waterjet process. This structure is designed to prevent the formation of blood pooling at the base of the wound. During wound packing, blood flow is reduced by compression. The perforated matrix allows excess blood to be guided along the wound channel into adjacent areas of the dressing. This promotes contact between blood and the distributed halloysite dots. By enabling interaction over a larger surface area, the design is intended to support clot formation along the wound channel. The aim is the development of a stable clot that contributes to maintaining hemostasis during patient transport.
Halloysite dots: The high hemostatic effectiveness of halloysite enables multifunctional applications in defined dot patterns rather than conventional full-surface coatings. Using a patented process, small, three-dimensional, lens-like structures are evenly applied across the carrier material. This design keeps the material flexible between the mineral dots, enabling it to adapt to the shape of the wound and providing good tactile control during packing. Meanwhile, the individual halloysite lenses resist compression, transferring externally applied pressure toward the wound bed, where it is needed most. Even when compressed, open spaces remain within the structure to allow excess blood to drain through the dressing. This combination of flexibility, pressure transmission, and an open structure reflects a targeted design approach developed specifically for effective wound packing in challenging situations.
Release behavior and printing technology: Hemostatic components that remain in the patient’s body are critical because they can lead to potential adverse effects such as thrombosis. During the development of SpeedM®, a patented coating technology was therefore developed that virtually eliminates the release of hemostatic material into the patient’s bloodstream. To monitor the safety and performance of the product, each production batch is tested by an independent institute for release behavior and efficacy in human citrate plasma.
